COVID-19 and Immune Dysregulation, a Summary and Resource

Highlights
1- COVID-19 causes turnover and aging of the immune cells responsible for responding to other pathogens. These cells have limitations in their proliferative capacity (ability to reproduce) and this gets used up when it is employed to generate cells for combating disease.
2- COVID-19 broadly activates these cells, so they have turnover – reproduce.
3- COVID-19 broadly activates these cells and thus ages them.
4- Naive T cells are relatively undifferentiated, and the stimulation will bring them more rapidly to terminal differentiation and reduce their proliferative capacity at the same time.
5- Studies also suggest SARS-COV-2 causes cell apoptosis (programmed cell death) of certain T cells in the immune system.
Viruses Affect the Immune System, Often Negatively
Many viruses can have devastating effects on the immune system, including measles, Epstein-Barr virus, HIV, influenza, and now SARS2. These effects can range from subtle to dramatic, making individuals more susceptible to other pathogens. Consequently, it is not surprising that SARS2 can cause harm to the immune system and increase susceptibility to other pathogens.
During the early phase of the SARS2 pandemic, infection was shown to result in severe depletion of T-cells in over 70% of non-ICU cases and 95% of those in ICU. There is some recovery over time, which takes months, but the long-term condition of the immune system often remains one of dysregulation—an important and multifaceted abnormal condition.
What is immune dysregulation and why is it important? Simply stated, the immune systems of patients with long COVID do not work properly. That gives them chronic symptoms and a risk of cancers, infections, and autoimmune disease. The level of this immune dysfunction is still in question, but many articles indicate that the effects are substantial.
Immune dysregulation is considered by many to be a manifestation of long COVID, but it occurs in patients after mild, moderate, and severe acute COVID-19. Long COVID is usually defined by symptoms, however, and immune dysregulation is often difficult to diagnose as a symptom. While its prevalence is still uncounted the association of immune dysregulation with long COVID indicates it is at least on the order of 10% and might be considerably larger.
The prevalence of acute COVID-19 infections has been incredibly high, resulting in far-reaching effects to humanity. Reinfections are becoming increasingly common, damaging the immune system, and leaving it weakened before subsequent infections occur. During this time, including the possibility of viral persistence with evidence from numerous studies, the immune system is not as strong and is more susceptible to other pathogens.
SARS-CoV-2 causes immune dysfunction through several direct and indirect mechanisms, including the killing of important categories of innate and adaptive immune cells. The rate of renewal of blood cells in an individual is a factor in determining how transient or severe the damage is.
The Immune System Is Not Like a Muscle, It Is Complex
The immune system consists of two parts: the innate immune system and the adaptive immune system. The innate system is more general and acts immediately in the case of a novel infection. The adaptive system is more specialized as it adapts to an infection to help ward off future infections (for good reading see here). The two systems work closely together. The immune system is amazingly complex consisting of T cells, B cells, antibodies, dendritic cells, platelets, and several more cell types. Each type of cell can have numerous subtypes.
T cells. T cells are one type of lymphocyte, a type of white blood cell, and are made in the bone marrow, maturing in the thymus gland. One of the functions of T cells is to carry out immune-mediated cell death. One of the two major subtypes of T cells, the cytotoxic T cell or CD8+ “killer cells,” attacks and destroys infected cells, cancer cells, and otherwise damaged cells. Another kind of T cells are the CD4+ “helper” T cells. These cells play an important role in the adaptive immune system. They aid the activity of other immune cells by releasing cytokines, a class of small proteins that aid in cell to cell signaling and are involved in inflammation. The CD8+ and CD4+ cells are named for proteins, CD8 or CD4, on the cell surface used for identification and binding. These cells can both release cytokines to recruit other types of cells when developing an immune response. The CD4+ T cells function as “helper cells.” These cells function by subsequent activation of memory B cells and the cytotoxic T cells, which then leads to a more pronounced immune response. T cells that are regulatory also exist. These prevent immune cells from reacting against one’s own cells in autoimmunity and thereby regulate the overall process. If this system breaks down, then, for example, regulatory T cells may be used by cancer cells to inhibit recognition of tumor cells by the other T cells, leading to cancer proliferation.
B cells. These cells are another type of lymphocyte, also white blood cells that mature in the bone marrow. B cells produce antibodies. Naive B cells are activated by molecules from an invading pathogen. Once activated, they proliferate and make the antibodies to help recognize the pathogen and allowing the immune system to destroy it.
Antibodies. Antibodies are large Y-shaped proteins, synthesized in the B cells, with two “sticking” ends that bind very selectively to molecules that have been identified by the B cells. With this binding mechanism, the antibody labels a pathogen or an infected cell by sticking to molecules on its surface. This in turn allows the killer T cells to identify these invaders for destruction. Antibodies can also stop pathogens or viruses directly by blocking part of an essential part of the invader, thereby making it inactive.
Dendritic cells. In the mammalian nervous system, dendritic cells work to present molecules on pathogenic invaders that killer cells and antibodies use to identify and then clear the pathogen. In this way they act as an intermediate between the innate and adaptive immune system. These cells grow long processes like the dendrites of nerve cells, hence the name, but are distinct from those cells. They are very important in directing the actions of immune defenders to the correct targets.
Monocytes. Another type of immune cells are monocytes. These are also white blood cells (leukocytes) found in the blood and tissues. The monocytes recruit other white blood cells in the quest to treat pathogenic injury and prevent infection. They are made in the bone marrow where they mature. Once mature, they can enter the bloodstream and tissues to defend your body against pathogens. The monocytes are stimulated to form two types of cells, the dendritic cells (see above) and macrophages. These latter cells are formed in tissues after leaving the blood and then can detect, engulf and destroy pathogens and apoptotic cells.
Platelets. These cells are an important part of blood clotting, or thrombosis, when vascular injury or opening occurs. As COVID-19 is a vascular disease, this is likely important. Think microclotting. However, they are also very important in the immune response and are often overlooked. Although thought of mainly for thrombosis, something called immuno-thrombosis is also initiated from the platelets by the innate immune system providing a critical first line of defense to locally control infection. They contain small granules (containers with nanometer dimension) that release serotonin, calcium, and adenosine phosphates to send messages when needed in the immune system.
COVID-19 Can Damage the Immune System
In the references attached we see that COVID-19 can affect and damage, at least temporarily, all the aspects and all the cell types of the immune system. It appears to age parts of the immune system, which might be permanent. We simply do not know yet. The effects of COVID-19 are seen in T cells, B cells, dendritic cells, monocytes, and platelets, among other systems.
COVID-19 causes turnover and aging of the immune cells responsible for responding to both COVID-19 and other pathogens. These cells have limitations in their proliferative capacity (ability to make new cells). Such a proliferative capacity is demonstrated by the Hayflick limit – the number of times a normal somatic, differentiated human cell population will divide before cell division stops, and this is not surprising as we know SARS-COV-2 also affects telomeres needed for proliferation.
COVID-19 infection broadly activates the immune cells, so they have greater turnover, which in turn ages them. Naive T cells are relatively undifferentiated, and the stimulation will bring them more rapidly to terminal differentiation and reduce their proliferative capacity. T cell production is also dependent on telomere length and these shorten naturally with age, hence the elderly are even more susceptible to telomere shortening brought on by COVID-19 infection.
Dendritic cells are instrumental in producing cytokines like interferon alpha, important in fighting viruses like RSV. Hence, a COVID-19-induced reduction of the plasmacytoid dendritic cell population might be, at least in part, responsible for RSV waves observed in many places.
Furthermore, even in mild COVID-19 infections, the monocytes have recently been shown to change their gene expression from a normal innate immune profile to a pro-thrombotic (promoting blood clotting) signature. This seems to be an important way that the immune system is compromised after infection and explains the clotting issues often observed even in mild COVID-19 patients.
Why Are Reinfections so Bad?
We know that reinfections occur, and they seem to be increasing. With our present high levels of transmission, many individuals will likely be experiencing two or more infections per year going forward. The immune system is damaged for more than half a year after infection, with dendritic cell deficiencies at least 7 months and both innate (NK cells, LD neutrophils, CXCR3+ monocytes) and adaptive immune cells (T helper, T follicular helper, and regulatory T cells) shown to be involved in immune dysfunction which had not ended at 8 months after infection. Reinfection has been shown to increase the cumulative risk of death, hospitalization, and sequelae in multiple organ systems in both the acute and postacute phase. This shows the immune system is not protective enough to limit damage by further COVID-19 infections or other infections, even after vaccination. This is a serious issue if we are continually reinfected before the immune system recovers each time. It is also important to remember that although children can still produce naive T cells, adults have a limited number. A very serious concern is that multiple COVID-19 infections early in life might age the immune system leading to immune systems similar to those of elderly in considerably younger individuals.
Our immunological protection against infection is also showing signs of decline with the omicron variant, and especially its new subvariants. Immune responses are not effectively induced against the omicron variant by previous infections with earlier variants, even in triply vaccinated individuals, and omicron infection itself is not an effective natural immune booster against itself. Furthermore, this study indicates a decline in the positive aspects of the immune response by some means. Hence, we can expect the negative aspects of infection to increase in severity because the antibody responses are not as effective. Combined with the increased SARS-COV-2 binding affinity, and reduced ability to clear the virus, it appears we should anticipate more chronic infections.
Summary
COVID-19 infection has several very concerning effects on the immune system that might easily lead to advantages for other pathogens as well as worse outcomes in COVID-19 reinfections. This immune dysfunction or aging is at least one of the most likely scenarios explaining the recent surges in diseases like RSV, influenza, Strep A, and other infections. By contrast, immunity debt is not considered a viable explanation as the surges of these diseases continue in countries that had surges last season and immunity wanes for many of these diseases. Another serious concern at this point is that repeated infections by COVID-19 might lead to depletion or exhaustion of cytotoxic CD8+ T cells which could have downstream effects on other diseases like cancers, as these T cells are instrumental in limiting tumor proliferation and have been shown to differentiate to become dysfunctional.
The following is a list of some of the research that has been carried out with relation to the effects of COVID-19 on the immune system.
READING LIST: SARS-COV-2/ COVID-19 Effects on the Immune System
(In chronological order)
March 19, 2020, Title: Functional exhaustion of antiviral lymphocytes in COVID-19 patients
“We showed that the total number of NK and CD8+ T cells was decreased markedly in patients with SARS-CoV-2 infection. The function of NK and CD8+ T cells was exhausted with the increased expression of NKG2A in COVID-19 patients. Importantly, in patients convalescing after therapy, the number of NK and CD8+ T cells was restored with reduced expression of NKG2A. These results suggest that the functional exhaustion of cytotoxic lymphocytes is associated with SRAS-CoV-2 infection. Hence, SARS-COV-2 infection may break down antiviral immunity at an early stage.”
https://www.nature.com/articles/s41423-020-0402-2
May 1 2020, Title: Reduction and Functional Exhaustion of T Cells in Patients With Coronavirus Disease 2019 (COVID-19)
”T cell counts are reduced significantly in COVID-19 patients, and the surviving T cells appear functionally exhausted.”
https://pubmed.ncbi.nlm.nih.gov/32425950/
July 2020, Title: AIDS and COVID-19 are two diseases separated by a common lymphocytopenia
”during the acute phase of their COVID-19 infection. A comparable reduction in B-cells was found in both diseases and a more severe reduction in the total amount of T-cells in COVID-19 compared to AIDS patients.”
August 13, 2020, Title: Long-term immunological health consequences of COVID-19
Good overview of basics. “The systemic COVID-19 syndrome is usually accompanied by mild lymphopaenia*, neutrophilia*, fever, thrombocytopenia*, hyperferritinaemia*, and elevation of other inflammatory and clotting factors.”
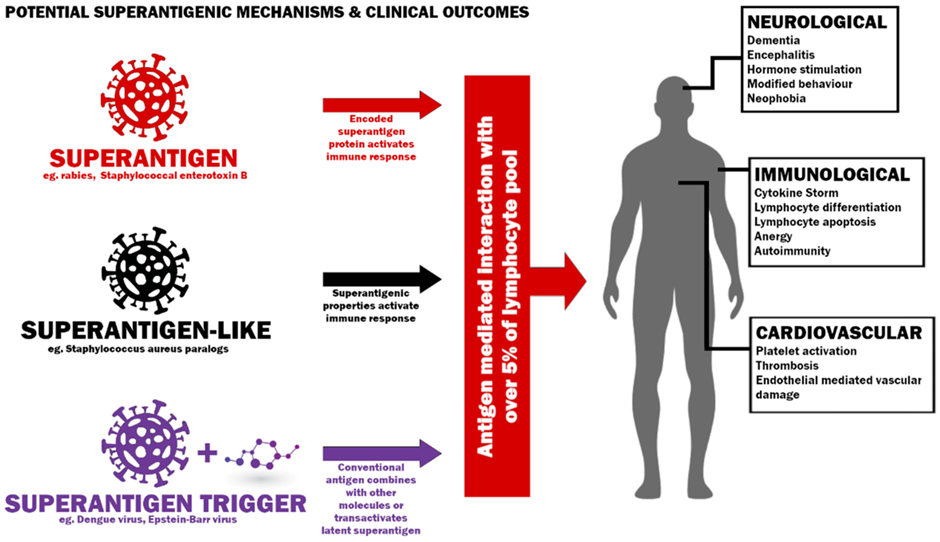
Sept 20 2020, Title: Superantigenic character of an insert unique to SARS-COV-2 spike supported by skewed TCR repertoire in patients with hyperinflammation
” superantigen-like motif is not present in other SARS family coronaviruses, which may explain the unique potential for SARS-COV-2 to cause both MIS-C and the cytokine storm observed in adult COVID-19.”
https://www.pnas.org/doi/10.1073/pnas.2010722117
September 28, 2020, Title: Immune dysfunction following COVID-19, especially in severe patients
“COVID-19 patients presented with dysregulated immune response (decreased T, B, and NK cells and increased inflammatory cytokines). Also, they were found to have increased levels of white blood cell, neutrophil count, and D-dimer in severe cases. Moreover, lymphocyte, CD4+ T cell, CD8+ T cell, NK cell, and B cell counts were lower in the severe group.”
https://www.nature.com/articles/s41598-020-72718-9
September 28 2020, Title: SARS-COV-2 Uses CD4 to Infect T Helper Lymphocytes
”SARS-COV-2 spike binds to CD4 and mediates entry of SARSCOV-2 in T cells, then assembles viral factories, impairs cell function and may cause cell death.” And “Here we show that SARS-COV-2 infects human CD4 + T helper cells, but not CD8 + T cells, and is present in blood and bronchoalveolar lavage T helper cells of severe COVID-19 patients. We demonstrated that SARS-COV-2 spike glycoprotein (S) directly binds to the CD4 molecule, which in turn mediates the entry of SARS-COV-2 in T helper cells in a mechanism that also requires ACE2 and TMPRSS2. Once inside T helper cells, SARS-COV-2 assembles viral factories, impairs cell function and may cause cell death. SARS-COV-2 infected T helper cells express higher amounts of IL-10, which is associated with viral persistence and disease severity. Thus, CD4-mediated SARS-CoV-2 infection of T helper cells may explain the poor adaptive immune response of many COVID-19 patients.” https://www.medrxiv.org/content/10.1101/2020.09.25.20200329v1.full.pdf
https://europepmc.org/article/PPR/PPR219346
Sept 28 2020, Title: Superantigenic character of an insert unique to SARS-CoV-2 spike supported by skewed TCR repertoire in patients with hyperinflammation
Superantigens in SARS-COV-2 spike protein in terms of MIS-C in children, suggests a possible role in the cytokine storm observed in adult patients. “Notably, the superantigen-like motif is not present in other SARS family coronaviruses, which may explain the unique potential for SARSCoV-2 to cause both MIS-C and the cytokine storm observed in adult COVID-19.”
https://doi.org/10.1073/pnas.2010722117
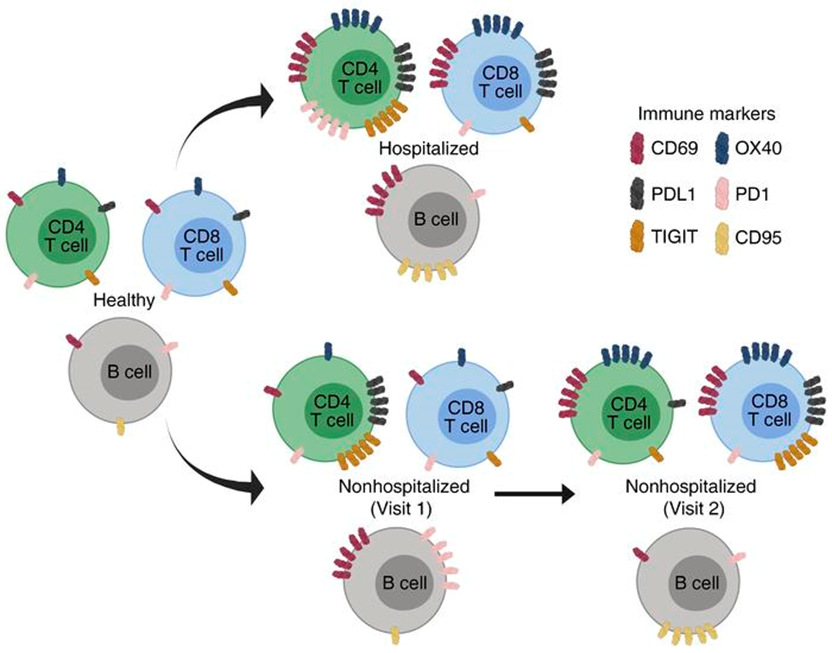
October 29, 2020, Title: Sustained cellular immune dysregulation in individuals recovering from SARS-CoV-2 infection
” Our findings show increased frequencies of T cell activation markers (CD69, OX40, HLA-DR, and CD154) in hospitalized patients, with other T cell activation/exhaustion markers (PD-L1 and TIGIT) remaining elevated in hospitalized and nonhospitalized individuals. B cells had a similar pattern of activation/exhaustion, with increased frequency of CD69 and CD95 during hospitalization followed by an increase in PD1 frequencies in nonhospitalized individuals. Interestingly, many of these changes were found to increase over time in nonhospitalized longitudinal samples, suggesting a prolonged period of immune dysregulation after SARS-CoV-2 infection.”
https://www.jci.org/articles/view/140491
Nov 17 2020, Title: Immune suppression in the early stage of COVID-19 disease
”We uncovered changes in the protein landscape that revealed immunosuppression and impaired tight junctions in COVID-19 patients in the early stage of infection.”
https://www.nature.com/articles/s41467-020-19706-9
December 15, 2020, Title: Sharing CD4+ T Cell Loss: When COVID-19 and HIV Collide on Immune System
“Both HIV-1 and SARS-CoV-2 infection share CD4+ T cell loss in association with disease outcome and immunodeficiency. Direct attacks on CD4+ T cells, immune activation and redistribution of CD4+ T cell are contributing mechanisms in very different proportion for CD4+ T cell lymphopenia in both diseases.”
https://www.frontiersin.org/articles/10.3389/fimmu.2020.596631/full
Dec 21, 2020, Title: Akt-Fas to Quell Aberrant T Cell Differentiation and Apoptosis in COVID-19
”depict SARS-Cov-2 as a lympho-manipulative pathogen; it distorts T cell function, numbers, and death, and creates a dysfunctional immune response”
https://www.frontiersin.org/articles/10.3389/fimmu.2020.600405/full
February 26, 2021, Title: COVID-19 Impairs Immune Response to Candida albicans
“COVID-19 patients displayed an immune phenotype characterized by increased HLA-DR+CD38+ and PD-1+ CD4+ and CD8+ T cells, and elevated CD8+CD244+ lymphocytes, compared to healthy controls. Monocyte activation markers and cytokines IL-6, IL-8, TNF, IL-10, and sIL2Rα were elevated, corresponding to monocyte activation syndrome, while IL-1β levels were low. LPS, HKLM and Aspergillus fumigatus antigen stimulation provoked an immune response that did not differ between COVID-19 patients and healthy controls, while COVID-19 patients showed an attenuated monocyte CD80 upregulation and abrogated release of IL-6, TNF, IL-1α, and IL-1β toward Candida albicans.”
https://www.frontiersin.org/articles/10.3389/fimmu.2021.640644/full
Mar 11 2021, Title, HLA class I–associated expansion of TRBV11-2 T cells in multisystem inflammatory syndrome in children.
This paper discusses superantigens in terms of MIS-C in children.
https://doi.org/10.1172/JCI146614.
April 22, 2021, Title: High-dimensional characterization of post-acute sequelae of COVID-19
A review with some discussion of viral persistence and immunity. “The mechanism or mechanisms that underlie the post-acute manifestations of COVID-19 are not entirely clear. Some of the manifestations may be driven by a direct effect of the viral infection, and may be explained by virus persisting in immune-privileged sites, an aberrant immune response, hyperactivation of the immune system or autoimmunity”
https://www.nature.com/articles/s41586-021-03553-9#MOESM4
May 25 2021, Title, Multisystem inflammatory syndrome in children is driven by zonulin-dependent loss of gut mucosal barrier.
This article again discusses superantigens in terms of MIS-C in children. This paper focusses on SARSCov2 in the GI tract of children promoting the superantigen in MIS-C.
https://doi.org/10.1172/JCI149633.
July 21, 2021, Title: Dendritic cell deficiencies persist seven months after SARS-CoV-2 infection
” Remarkably, previously hospitalized and nonhospitalized patients remained with decreased numbers of CD1c+ myeloid DCs and pDCs seven months after SARS-CoV-2 infection. Moreover, the expression of DC markers such as CD86 and CD4 were only restored in previously nonhospitalized patients, while no restoration of integrin β7 and indoleamine 2,3-dyoxigenase (IDO) levels were observed. These findings contribute to a better understanding of the immunological sequelae of COVID-19.” And “In summary, we have demonstrated that SARS-CoV-2 infected patients showed a deficit in some DC subsets and alterations in DC homing and activation markers, which are not restored more than 7 months after the infection independently of previous hospitalization. Our results suggest that there is an ongoing inflammation which could be partially induced by DCs, these findings might contribute to a better understanding of the immunological sequelae of COVID-19.”
https://www.nature.com/articles/s41423-021-00728-2
Sept 22 2021, Title: SARS-CoV-2 infection causes immunodeficiency in recovered patients by downregulating CD19 expression in B cells via enhancing B-cell metabolism
”In conclusion, we found that SARS-CoV-2 infection causes immunodeficiency in recovered patients by downregulating CD19 expression in B cells via enhancing B-cell metabolism”
https://www.nature.com/articles/s41392-021-00749-3
Oct 1 2021, Title: Uncertainty around the Long-Term Implications of COVID-19
”a perspective on some of the long-term risks of mutational escape, viral persistence, reinfection, immune dysregulation and neurological and multi-system complications (Long COVID).”
https://www.mdpi.com/2076-0817/10/10/1267/htm
October 28, 2021, Title: How COVID-19 alters the immune system
“The SARS-CoV-2 coronavirus causes moderate to severe disease in 3-10% of those infected. In such cases, the immune system overreacts to the virus, triggering an aberrant innate immune response that is characterized by systemic inflammation, intravascular blood clotting and damage to the cardiovascular system.”
https://www.sciencedaily.com/releases/2021/10/211028125803.htm
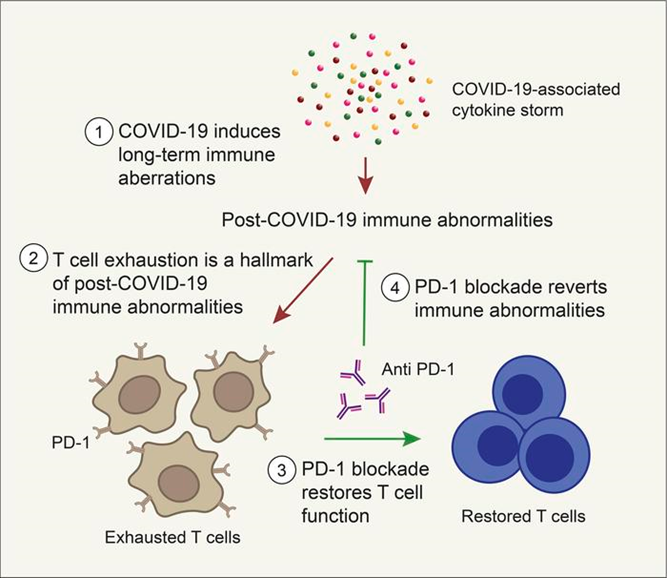
Nov 16 2021, Title: PD-1 blockade counteracts post–COVID-19 immune abnormalities and stimulates the anti–SARS-CoV-2 immune response
“We extensively immunologically characterized patients who recovered from COVID-19. In these patients, T cells were exhausted, with increased PD-1+ T cells, as compared with healthy controls. Plasma levels of IL-1β, IL-1RA, and IL-8, among others, were also increased in patients who recovered from COVID-19. This altered immunophenotype was mirrored by a reduced ex vivo T cell response to both nonspecific and specific stimulation, revealing a dysfunctional status of T cells, including a poor response to SARS-CoV-2 antigens.”
https://insight.jci.org/articles/view/146701
January 14, 2022, Title: Long-term perturbation of the peripheral immune system months after SARS-CoV-2 infection
“Deep immunophenotyping revealed significant differences in multiple innate (NK cells, LD neutrophils, CXCR3+ monocytes) and adaptive immune populations (T helper, T follicular helper, and regulatory T cells) in convalescent individuals compared to healthy controls, which were most strongly evident at 12 and 16 wpi. RNA sequencing revealed significant perturbations to gene expression in COVID-19 convalescents until at least 6 months post-infection. We also uncovered significant differences in the transcriptome at 24 wpi of convalescents who were referred to a long COVID clinic compared to those who were not.”
https://bmcmedicine.biomedcentral.com/articles/10.1186/s12916-021-02228-6
Jan 22 2022, Title: Immunological dysfunction persists for 8 months following initial mild-
to-moderate SARS-CoV-2 infection
Important paper showing immune function was not restored even at 8 months.
https://www.nature.com/articles/s41590-021-01113-x?s=09
Jan 22 2022, Title: T cell apoptosis characterizes severe Covid-19 disease
”results are compatible with a model in which T-cell apoptosis accounts for T lymphopenia in individuals with severe COVID-19. Therefore, a strategy aimed at blocking caspase activation could be beneficial for preventing immunodeficiency in COVID-19 patients.”
https://www.nature.com/articles/s41418-022-00936-x
February 1, 2022, Title: Immune response pattern across the asymptomatic, symptomatic and convalescent periods of COVID-19
“The immune response is activated in asymptomatic infection, but defective in mild and severe patients, in which neutrophils were involved.
Disorders of immune responses and cholesterol metabolism span the entire progression of the disease until recovery.”
https://www.sciencedirect.com/science/article/pii/S1570963921001424?via%3Dihub
February 21, 2022, Title: Depletion and Dysfunction of Dendritic Cells: Understanding SARS-CoV-2 Infection
“Dendritic cells (DCs) “bridge” innate immunity and adaptive immunity. … We also discuss the decline in DC number, the impaired antigen-presentation capability, and the reduced production of type-I interferon of DCs in severe SARS-CoV-2 infection.”
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8898834/
March 3 2022, Title: Understanding the Effects of Age and T-Cell Differentiation on COVID-19 Severity: Implicating a Fas/FasL-mediated Feed-Forward Controller of T-Cell Differentiation. This opinion article concludes, “T-cell differentiation and acquisition of effector function is accomplished via Feed-forward control.” A mechanism for the superantigen in the deterioration of immune response in COVID-19.
https://www.frontiersin.org/articles/10.3389/fimmu.2022.853606/full
March 10 2022, Title: The immunology and immunopathology of COVID-19
Cautious overview. 1 mention of t cell lowering in long covid.
https://www.science.org/doi/10.1126/science.abm8108
March 11, 2022, Title: ACE2-independent infection of T lymphocytes by SARS-CoV-2
”Here, we showed that SARS-CoV-2 infected T lymphocytes, mainly CD4 + T cells, in an ACE2-independent manner. SARS-CoV-2 infection triggered pronounced T-cell death, which potentially contributed to lymphopenia in patients with COVID-19.”
https://www.nature.com/articles/s41392-022-00919-x#citeas
March 23 2022, Title: Superantigens and SARS-CoV-2
Superantigen defines ”proteins that hyper-stimulate T cells via the crosslinking of T cell receptors (TCR) with MHC Class II molecules”
https://www.mdpi.com/2076-0817/11/4/390/htm
April 6, 2022, Title: FcγR-mediated SARS-CoV-2 infection of monocytes activates inflammation
“Taken together, these findings suggest that antibody-mediated SARS-CoV-2 uptake by monocytes and macrophages triggers inflammatory cell death that aborts the production of infectious virus but causes systemic inflammation that contributes to COVID-19 pathogenesis.”
https://www.nature.com/articles/s41586-022-04702-4
April 8 2022, Title: SARS-CoV-2 and its variants, but not Omicron, induces thymic atrophy and impaired 2 T cell development
”first report of SARS-CoV-2 46 associated thymic atrophy resulting from impaired T cell developmental pathway and also 47 explains dysregulated T cell function in COVID-19”
https://www.biorxiv.org/content/10.1101/2022.04.07.487556v1.full.pdf
April 14 2022, Title: SARS-CoV-2 Actively Infects And Kills Lymphoid Cells
Kills T cells.
“In a recent paper published by the journal Nature, scientists describe a new target for the SARS-CoV-2 virus that is independent of ACE2 receptors—our T cells.”
Paper here (see later): https://www.nature.com/articles/s41392-022-00919-x#citeas
April 19, 2022, Title: Accelerated biological aging in COVID-19 patients
Indirectly related to the immune system. “We also find the increasing acceleration of epigenetic aging and telomere attrition in the sequential blood samples from healthy individuals and infected patients developing non-severe and severe COVID-19. In addition, the longitudinal DNA methylation profiling analysis find that the accumulation of epigenetic aging from COVID-19 syndrome could be partly reversed at late clinic phases in some patients.”
https://www.nature.com/articles/s41467-022-29801-8?s=09
April 22, 2022, Title: Reduced Cell Surface Levels of C-C Chemokine Receptor 5 and Immunosuppression in Long Coronavirus Disease 2019 Syndrome
““Long COVID” is characterized by chronic and often debilitating symptoms following acute coronavirus disease 2019 (COVID-19). These findings suggest an unexpected mechanism of abnormal immune downmodulation in some persons that is normalized by leronlimab.”
https://academic.oup.com/cid/article/75/7/1232/6572226
May 7, 2021, Title: Longitudinal Analysis of COVID-19 Patients Shows Age-Associated T Cell Changes Independent of Ongoing Ill-Health
“We demonstrate myeloid recovery but persistent T cell abnormalities in convalescent COVID-19 patients more than three months after initial infection. These changes are more marked with age and are independent of ongoing subjective ill-health, fatigue and reduced exercise tolerance.”
https://www.frontiersin.org/articles/10.3389/fimmu.2021.676932/full
May 14 2022, Title: T-cell recovery and evidence of persistent immune activation 12 months after severe COVID-19
”T-cell numbers and function recover in most patients after COVID-19. However, we find evidence of persistent T-cell activation up to 12 months after infection and describe a subgroup of severe COVID-19 patients with persistently low CD8+ T-cell counts exhibiting a dysregulated immune response during acute infection.”
https://onlinelibrary.wiley.com/doi/10.1111/all.15372
May 16, 2022, Title: SARS-CoV-2 variants do not evolve to promote further escape from MHC-I recognition
“VOCs retain similar MHC-I downregulation capacity compared to the ancestral virus. However, VOCs exhibit a greater ability to suppress type I IFN than the ancestral virus. Although VOCs possess unique mutations within the ORF8 gene, which suppresses MHC-I expression, none of these mutations enhanced the ability of ORF8 to suppress MHC-I expression.” CD8 T cells help fight off viral infection by detecting and killing infected cells. CD8 T cells detect MHC I plus the viral peptide on infected cells. One of the ways viruses like SARS-COV-2 use to avoid killing is to inhibit MHC-I expression and presentation on the cells.
https://www.biorxiv.org/content/10.1101/2022.05.04.490614v2
May 18, 2022, Title: Unexplained post-acute infection syndromes
Good review of post-acute infection syndromes including the immune system and COVID-19.
https://www.nature.com/articles/s41591-022-01810-6

May 20 2022 -> November 15, 2022, Title: SARS-CoV-2 Spike protein suppresses CTL-mediated killing by inhibiting immune synapse assembly
”These results highlight a new strategy of immune evasion by SARS-CoV-2 based on the Spike-dependent, ACE2-mediated targeting of the lytic IS to prevent elimination of infected cells.”
June 16, 2022, Title: Infection of lung megakaryocytes and platelets by SARS-CoV-2 anticipate fatal COVID-19
“In sum, the presence of infectious SARS-CoV-2 in platelets combined with the intricate relationship between hemostasis, inflammation and the spread of infection has major consequences on COVID-19 pathogenesis and can turn out fatal. Anti-platelet drugs might be explored to develop anti-inflammatory coupled to anti-thrombotic treatment against severe SARS-CoV-2.”
https://link.springer.com/article/10.1007/s00018-022-04318-x
July 28, 2022, Title: Impaired ketogenesis ties metabolism to T cell dysfunction in COVID-19
“In COVID-19 acute respiratory distress syndrome (ARDS), T cells are exhausted and skewed towards glycolysis, probably as a result of reduced oxygenation of the pulmonary tissue. β-hydroxybutyrate (BHB) not only rewires T cells to OXPHOS by fuelling the TCA cycle, but also promotes the synthesis of amino acids that are depleted in COVID-19 acute respiratory distress syndrome. Consequently, impaired ketogenesis and production of BHB in COVID-19 ARDS may be at the root of the metabolic dysregulation and defective effector function of T cells, and may predispose patients to pulmonary pathology, including fibrosis.”
https://www.nature.com/articles/s41586-022-05128-8
August 1, 2022, Title: Postacute COVID-19 is Characterized by Gut Viral Antigen Persistence in Inflammatory Bowel Diseases
Viral persistence. ”Our results indicate that SARS-CoV-2 antigen persistence in infected tissues serves as a basis for postacute COVID-19. The concept that viral antigen persistence instigates immune perturbation and postacute COVID-19 requires validation in controlled clinical trials.”
https://www.gastrojournal.org/article/S0016-5085(22)00450-4/fulltext
Good editorial here: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9175081/
Aug 19, 2022, Title: Single-cell RNA sequencing highlights a reduced function of natural killer and cytotoxic T cell in recovered COVID-19 pregnant women
”CD8+ 67 68 cytotoxic T cells were compromised in recovered patients compared with healthy 69 pregnant women”
https://www.biorxiv.org/content/10.1101/2022.08.18.504053v1.full.pdf
Aug 10 2022, Title: Distinguishing features of Long COVID identified through immune profiling
“provides a basis for future investigations into the immunological underpinnings driving the genesis of Long COVID.”
https://www.medrxiv.org/content/10.1101/2022.08.09.22278592v1.full.pdf
August 16, 2022, Title: Structural epitope profiling identifies antibodies associated with critical COVID-19 and long COVID
”We identify patterns of epitope-binding antibodies associated with immunopathology, including a non-isotype switching IgM response to a membrane protein epitope which is the strongest single immunological feature associated with severe COVID-19 to date (adjusted OR 72.14, 95% CI: 9.71 – 1300.15). We suggest the mechanism is T independent B cell activation and identify persistence (> 1 year) of this response in individuals with long COVID particularly affected by fatigue and depression.”
https://www.medrxiv.org/content/10.1101/2022.07.11.22277368v2
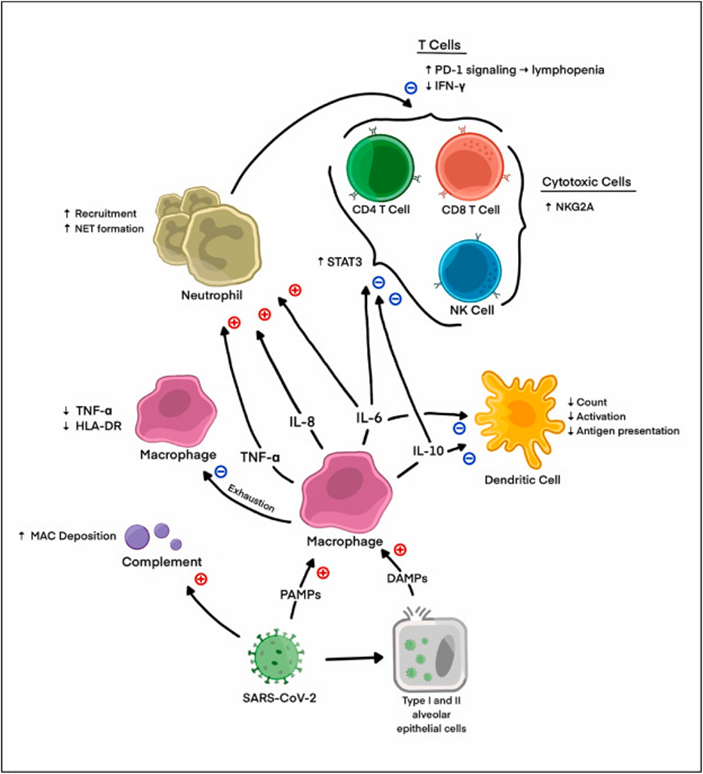
September, 2022, Title: COVID-19 disease and immune dysregulation
“Given its severity and complexity, we performed a narrative literature review to evaluate the mechanisms of immune dysregulation that is specific to cell type, disease, and individual immune systems to unlock targeted therapies for COVID-19 disease and identify future paths for immunologic research.” Review of COVID-19 induced dysregulation by immune cell type focusing on severe and acute disease, but presenting data on continued dysfunction and potential treatments and tests.”
https://www.sciencedirect.com/science/article/pii/S1521692622000561
September 1, 2022, Title: Chronic active Epstein-Barr exacerbated by COVID-19 co-infection
Case study, “A COVID-19 co-infection (with Epstein Barr) resulted in rapid health decline and death.”
https://www.ijidonline.com/article/S1201-9712(22)00439-8/fulltext
September 2, 2022, Title: Mucosal Immunity After Novel COVID-19 Infection – Virus-Induced Immunosuppression: Preliminary Study
“In recovered COVID-19 patients, the cell-bound component of mucosal immunity reveals lympho- and eosinopenia. The neutrophils population shows cytodestruction, increased apoptosis, and decreased functional and metabolic activity of the cell.”
https://link.springer.com/article/10.1007/s12668-022-01020-x
September 26, 2022, Title: Impact of cross-coronavirus immunity in post-acute sequelae of COVID-19
”A distinct humoral immune response was observed in individuals with PASC. Specifically, individuals with PASC harbored less inflamed and weaker Fcγ receptor binding anti-SARS-CoV-2 antibodies and a significantly expanded and more inflamed antibody response against endemic Coronavirus OC43. These findings implicate previous common Coronavirus imprinting as a marker for the development of PASC.”
https://www.medrxiv.org/content/10.1101/2022.09.25.22280335v1
Oct 1 2022, Title: Reduced Cell Surface Levels of C-C Chemokine Receptor 5 and Immunosuppression in Long Coronavirus Disease 2019 Syndrome
These findings suggest an unexpected mechanism of abnormal immune downmodulation in some persons that is normalized by leronlimab.
https://academic.oup.com/cid/article/75/7/1232/6572226
Oct 19 2022, Title: Impact of SARS-CoV-2 ORF6 and its variant polymorphisms on host responses and viral pathogenesis
” ORF6 as a key SARS-CoV-2 innate immune antagonist”
https://www.biorxiv.org/content/10.1101/2022.10.18.512708v1.full.pdf
October 19, 2022, Title: Tissue Injury and Leukocyte Changes in Post-Acute Sequelae of SARS-CoV-2: Review of 2833 Post-acute Patient Outcomes per Immune Dysregulation and Microbial Translocation in Long COVID
“Taken together, we develop the premise that SARS-CoV-2 infection results in PASC as a consequence of acute and/or persistent single or multiple organ injury mediated by PASC determinants to include degree of host response (inflammation, NETinjury), residual viral antigen (persistent antigen) and exogenous factors (microbial translocation).”
https://www.preprints.org/manuscript/202210.0342/v1
October 22, 2022, Title: Impact of the COVID-19 pandemic on the circulation of other pathogens in England
“Despite concerns that a lack of exposure to common pathogens may affect population immunity and result in large outbreaks by various pathogens post-COVID-19, only four of the 22 investigated diseases and disease groups displayed higher post-than pre-pandemic levels without an obvious causative relationship.”
https://www.medrxiv.org/content/10.1101/2022.10.21.22281366v1
Oct 24 2022, Title: In Cleveland and beyond researchers begin to unravel the mystery of long COVID-19
Covid similar in some ways to AIDS
November 1, 2022, Title: Gut microbiome dysbiosis in antibiotic-treated COVID-19 patients is associated with microbial translocation and bacteremia
” Analysis of blood culture results testing for secondary microbial bloodstream infections with paired microbiome data indicates that bacteria may translocate from the gut into the systemic circulation of COVID-19 patients. These results are consistent with a direct role for gut microbiome dysbiosis in enabling dangerous secondary infections during COVID-19.”
https://www.nature.com/articles/s41467-022-33395-6
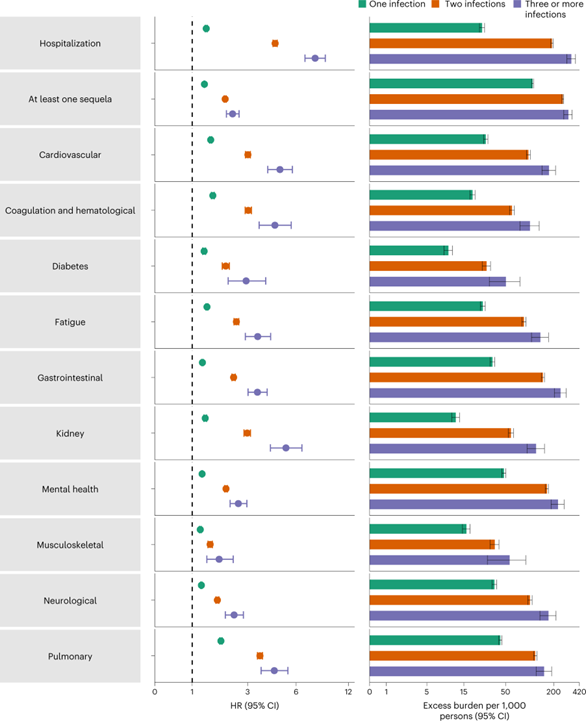
November 10, 2022, Title: Acute and postacute sequelae associated with SARS-CoV-2 reinfection
“The evidence shows that reinfection further increases risks of death, hospitalization and sequelae in multiple organ systems in the acute and postacute phase.” This suggests the immune system is not protective enough to limit damage with further infections.
https://www.nature.com/articles/s41591-022-02051-3
“Together, our findings indicate that platelet activation and hypercoagulable state persists in COVID-19 survivors after hospital discharge, having possible implications for long lasting consequences of COVID-19.”
November 11, 2022, Title: Persisting Platelet Activation and Hyperactivity in COVID-19 Survivors
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9645447/
November 15, 2022, Title: Things everyone should know about COVID-19 (Part 1: Immune System) by Jeff Gilchrist, “Did you know that the COVID-19 virus (SARS-CoV-2) actually has the ability to actively suppress and hide from multiple parts of your immune system?” Excellent layperson overview.
November 20, 2022, Title: Elevated circulating monocytes and monocyte activation in pulmonary post-acute sequelae of SARS-CoV-2 infection
“Monocytes and macrophages play a pivotal role in inflammation during acute SARS-CoV-2 infection…. Conclusion: This study presents evidence that COVID convalescents exhibit monocyte alteration beyond the acute COVID-19 infection period even in convalescents with no residual symptoms.”
https://www.medrxiv.org/content/10.1101/2022.11.19.22282543v1
November 23, 2022, Title: Direct Cryo-ET observation of platelet deformation induced by SARS-CoV-2 Spike protein
”Live imaging showed that the S protein triggers platelets to deform dynamically, in some cases, leading to their irreversible activation. Strikingly, cellular cryo-electron tomography revealed dense decorations of S protein on the platelet surface, inducing filopodia formation.”
https://www.biorxiv.org/content/10.1101/2022.11.22.517574v1
November 27, 2022, Title: Prolonged T-cell activation and long COVID symptoms independently associate with severe disease at 3 months in a UK cohort of hospitalized COVID-19 patients
”In summary, our study highlights a complex recovery of the immune system following severe COVID-19 with evidence of persistent activation of CD4+ and CD8+ 439 T-cells, which may be bystander driven, and 440 elevated levels of T-cell-related cytokines in the plasma of severe patients at 3 months. Our data suggests the lack of a direct association between long COVID and immune activation markers and pro-inflammatory cytokines measured in this study.”
https://www.medrxiv.org/content/10.1101/2022.11.25.22282759v1.full.pdf
November 29, 2022; Title: Disruption in seasonality, patient characteristics and disparities of respiratory syncytial virus infection among young children in the US during and before the COVID-19 pandemic: 2010-2022
“Among RSV-infected children in 2022, 19.2% had prior documented COVID-19 infection, significantly higher than the 9.7% among uninfected children, suggesting that prior COVID-19 could be a risk factor for RSV infection or that there are common risk factors for both viral infections.”
https://www.medrxiv.org/content/10.1101/2022.11.29.22282887v1
November-December, 2022, Title: Asymptomatic SARS-COV-2 infection in children’s tonsils
”Positive immunostaining in adenotonsillar tissue samples suggest that lymphoid tissue can be a reservoir of SARS-CoV-2 and may play an important role in community dissemination. It remains unclear for how long the lymphoid tissue can sustain the SARS-CoV-2 in a persistent infection, and whether this persistence has any impact on virus transmission.”
https://www.sciencedirect.com/science/article/pii/S1808869422001392
December 2, 2022, Title: Single-cell multiomics revealed the dynamics of antigen presentation, immune response and T cell activation in the COVID-19 positive and recovered individuals
”Importantly, we discovered that a subset of the naive T-cells from the healthy individuals were absent from the recovered individuals, suggesting a post-infection inflammatory stage. Both COVID-19 positive patients and the recovered individuals exhibited a CD40-CD40LG-mediated inflammatory response in the monocytes and T-cell subsets. T-cells, NK-cells, and monocyte-mediated elevation of immunological, stress and antiviral responses were also seen in the COVID-19 positive and the recovered individuals, along with an abnormal T-cell activation, inflammatory response, and faster cellular transition of T cell subtypes in the COVID-19 patients.”
https://www.frontiersin.org/articles/10.3389/fimmu.2022.1034159/full
December 7, 2022, Title: Increased migratory/activated CD8+ T cell and low avidity SARS-CoV-2 reactive cellular response in post-acute COVID-19 syndrome
“The phenotyping of lymphocytes showed a significantly higher number of CD8+ T cells expressing the Epstein-Barr virus induced G protein coupled receptor 2, chemokine receptor CXCR3 and C-C chemokine receptor type 5 playing an important role in inflammation and migration in PASC patients compared to controls. Additionally, a stronger, SARS-CoV-2 reactive CD8+ T cell response, characterized by IFNγ production and predominant TEMRA phenotype but low SARS-CoV-2 avidity was detected in PASC patients compared to controls. Furthermore, higher titers of several autoantibodies were detected among PASC patients. Our data suggest that a persistent inflammatory response triggered by SARS-CoV-2 might be responsible for the observed sequelae in PASC patients.”
https://www.biorxiv.org/content/10.1101/2022.12.03.519007v1
December 8, 2022, Title: Mild SARS-CoV-2 infection results in long-lasting microbiota instability
“SARS-CoV-2 cases exhibited a significantly less stable gut microbiota relative to controls, as long as 154 days after their positive test. These results were confirmed and extended in the K18-hACE2 mouse model, which is susceptible to SARS-CoV-2 infection. All of the tested SARS-CoV-2 variants significantly disrupted the mouse gut microbiota, including USA-WA1/2020 (the original variant detected in the United States), Delta, and Omicron.” And “SARS-CoV-2 most likely impacts the gut microbiome through effects on host immune or epithelial cells.”
https://www.biorxiv.org/content/10.1101/2022.12.07.519508v1.full.pdf
December 15, 2022, Title: Longitudinal Analysis of T cells in COVID-19 Survivors with Post-Acute Sequelae of COVID-19 Reveals Associations between Individual Symptoms and Inflammatory Indexes
“These findings suggest continued CD8+ T cell and CD4+CD25+ T cell activation and response following SARS-CoV-2 infection in patients with PASC. An increase in T regulatory cells suggests an ongoing attempt to control host inflammation in a subset of these patients. These results shed further light on continued immune system activation and chronic inflammation as a link to symptoms in COVID-19 survivors suffering from PASC.”
https://academic.oup.com/ofid/article/9/Supplement_2/ofac492.003/6902318?login=false
December 23, 2022, Title: SARS-CoV-2 Omicron subvariants evolved to promote further escape from MHC-I recognition
“Notably, MHC-I upregulation was strongly inhibited after SARS-CoV-2 infection in vivo. While earlier VOCs possess similar capacity as the ancestral strain to suppress MHC I, Omicron subvariants exhibit a greater ability to suppress surface MHC-I expressions. Collectively, our data suggest that, in addition to escape from neutralizing antibodies, the success of Omicron subvariants to cause breakthrough infection and reinfection may in part be due to its optimized evasion from T cell recognition.”
Omicron variants further suppress MHC I expression. MHC is a molecular identifier for killer T cells to work.
https://www.biorxiv.org/content/10.1101/2022.05.04.490614v3.full.pdf
December 26, 2022, Title: Transcriptional reprogramming from innate immune functions to a pro-thrombotic signature by monocytes in COVID-19
“Monocytes from COVID-19 patients display altered expression of cell surface receptors and a dysfunctional metabolic profile that distinguish them from healthy monocytes. Secondary pathogen sensing ex vivo leads to defects in pro-inflammatory cytokine and type-I IFN production in moderate COVID-19 cases, together with defects in glycolysis. COVID-19 monocytes switch their gene expression profile from canonical innate immune to pro-thrombotic signatures and are functionally pro-thrombotic, both at baseline and following ex vivo stimulation with SARS-CoV-2. Transcriptionally, COVID-19 monocytes are characterized by enrichment of pathways involved in hemostasis, immunothrombosis, platelet aggregation and other accessory pathways to platelet activation and clot formation. These results identify a potential mechanism by which monocyte dysfunction may contribute to COVID-19 pathology.” Clearly reminds us we are more vulnerable during recovery from SARS-COV-2 infection.
https://www.nature.com/articles/s41467-022-35638-y
January 11, 2023, Title: A single-cell atlas reveals shared and distinct immune responses and metabolism during SARS-CoV-2 and HIV-1 infections
” Highlights
- COVID-19 and HIV-1+ patients show disease-specific inflammatory immune signatures
- COVID-19 patients show more productive humoral responses than HIV-1+ patients
- SARS-CoV-2 elicits more enriched IFN-I signaling relative to HIV-I
- Divergent, impaired metabolic programs distinguish SARS-CoV-2 and HIV-1 infections”
https://www.biorxiv.org/content/10.1101/2022.01.10.475725v1
January 14, 2023, Title: Long-term perturbation of the peripheral immune system months after SARS-CoV-2 infection
”In conclusion, this study found persistent changes to the peripheral immune system of SARS-CoV-2 convalescents until at least 6 months post-infection and identified a subset of these changes that were associated with long COVID. These changes to the peripheral immune system could have implications for how individuals recovering from SARS-CoV-2 infection respond to other infections encountered in this period and persistent immune activation may also exacerbate other chronic conditions.”
https://bmcmedicine.biomedcentral.com/articles/10.1186/s12916-021-02228-6
January 18, 2023, Title: COVID-19 INDUCES SENESCENCE AND EXHAUSTION OF T CELLS IN PATIENTS WITH MILD/MODERATE AND SEVERE DISEASE DURING A SEVEN-DAY INTERVAL
Small sample cohort. ”Our longitudinal study provided evidence of accelerated immunosenescence in the T cell compartment during COVID-19 showing that senescent and exhausted T cells significantly increased within a seven-day period. Interestingly, even though our samples were composed by two different populations, who presented distinct inflammatory profiles and were possibly affected by different variants of the virus, a similar profile of immunossenescence and exhaustion was identified during this short period of infection by SARS-CoV-2. Furthermore, this change in immunological profile was shown to be independent of disease severity even though the magnitude was greater in patients with severe disease. This suggests that accelerated immunosenescence of the T cell compartment may be described as a general feature of COVID-19.”
https://www.medrxiv.org/content/10.1101/2023.01.16.23284612v1
January 18, 2023, Title: How your first brush with COVID warps your immunity
”The immune system responds more strongly to the strain of a virus that it first met, weakening response to other strains. Can this ‘imprinting’ be overcome?”
https://www.nature.com/articles/d41586-023-00086-1
January 19, 2023, Title: Alterations in platelet proteome signature and impaired platelet integrin αIIbβ3 activation in patients with COVID-19
”Overall, our data suggest the presence of 2 platelet populations in patients with COVID-19. The first is circulating platelets with an altered proteome, increased basal PS exposure, and reduced agonist– induced integrin αIIbβ3 activation. The second platelet population is P-selectin expressing neutrophil–associated platelets. Furthermore, circulating platelets from COVID-19 patients have a unique protein signature, with multiple COVID-19 associated plasma proteins being markedly enhanced. Our data show a complex picture and suggest that platelet–driven thromboinflammation may be one of the key drivers enhancing the risk of thrombosis in COVID-19 patients. The data also point toward potential mechanisms of this effect, which now need to be further characterized.”
https://www.jthjournal.org/article/S1538-7836(23)00059-4/pdf
February 2023, Title: Risk of autoimmune diseases in patients with COVID-19: A retrospective cohort study
”In conclusion, our preliminary data suggest that COVID-19 is associated with a significantly different risk of various autoimmune diseases. It is crucial for physicians to have relevant notions and to recognize these autoimmune manifestations in order to respond appropriately in the ongoing pandemic and long-term post-pandemic phase. The impact of vaccination on the development of autoimmune diseases should also be studied in the future.”
https://www.sciencedirect.com/science/article/pii/S2589537022005120

February 4, 2023, Title: Direct Cryo-ET observation of platelet deformation induced by SARS-CoV-2 spike protein
”Here, we report the direct observation of the interactions between S protein and platelets. Live imaging shows that the S protein triggers platelets to deform dynamically, in some cases, leading to their irreversible activation. Cellular cryo-electron tomography reveals dense decorations of S protein on the platelet surface, inducing filopodia formation.”
https://www.nature.com/articles/s41467-023-36279-5
February 10, 2023, Title: SARS-CoV-2 NSP5 Antagonizes the MHC II Antigen Presentation Pathway by Hijacking Histone Deacetylase 2
”These results identify a novel mechanism by which SARS-CoV-2 can limit antigen presentation on MHC II, thereby delaying or weakening the subsequent adaptive immune response.”
https://www.biorxiv.org/content/10.1101/2023.02.10.528032v1
February 19, 2023, Title: Long COVID manifests with T cell dysregulation, inflammation, and an uncoordinated adaptive immune response to SARS-CoV-2
“Our findings demonstrate that individuals with LC exhibit systemic inflammation and immune dysregulation. This is evidenced by global differences in T cell subset distribution in ways that imply ongoing immune responses, as well as by sex-specific perturbations in cytolytic subsets. Individuals with LC harbored increased frequencies of CD4+ T cells poised to migrate to inflamed tissues, and exhausted SARS-CoV-2-specific CD8+ T cells. They also harbored significantly higher levels of SARS-CoV2 antibodies, and in contrast to non-LC individuals, exhibited a mis-coordination between their SARS-CoV-2-specific T and B cell responses. Collectively, our data suggest that proper crosstalk between the humoral and cellular arms of adaptive immunity has broken down in LC, and that this, perhaps in the context of persistent virus, leads to the immune dysregulation, inflammation, and clinical symptoms associated with this debilitating condition.”
https://www.biorxiv.org/content/10.1101/2023.02.09.527892v1.full.pdf


